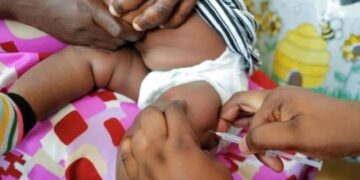
The world’s first malaria treatment specifically designed for babies has been approved, in what health experts are calling a major step forward in the fight against one of Africa’s deadliest diseases.
The World Health Organization has granted prequalification to Coartem Baby, meaning it meets global standards for safety, quality and effectiveness and can now be widely procured and distributed, especially across malaria-endemic regions.
Filling a long-standing treatment gap
Until now, infants under six months had no dedicated malaria treatment. Doctors often relied on drugs designed for older children, increasing the risk of incorrect dosing and harmful side effects.
This gap has been critical. Malaria killed about 610,000 people in 2024, with roughly three-quarters of those deaths occurring among children under five in Africa.
Health experts say up to 18% of babies under six months in parts of the continent are infected with malaria, despite earlier assumptions that maternal immunity offered protection.
A drug designed for the smallest patients
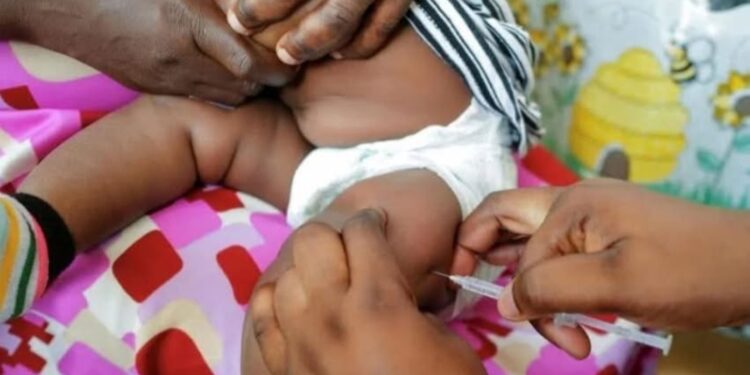
Coartem Baby is formulated for infants weighing as little as 2kg. It comes as sweet, cherry-flavoured tablets that dissolve easily in liquids, including breast milk, making it easier for caregivers to administer.
The treatment combines two established antimalarial drugs and was developed by Novartis in partnership with Medicines for Malaria Venture.
Dr Tedros Adhanom Ghebreyesus said the approval marks a turning point in a long battle.
“For centuries, malaria has taken children’s lives and weakened communities. Today, that story is starting to change,” he said.
Early rollout and real-life impact
The treatment has already been introduced in Ghana, where doctors are seeing early results.
At a hospital in Ankaase, a baby diagnosed with malaria at just 12 weeks old was successfully treated with the drug and has since recovered.
Doctors say the availability of a treatment tailored for infants gives them more confidence in diagnosing and managing malaria in very young patients.
What it means for Africa
The approval is expected to significantly improve survival rates among infants, particularly in sub-Saharan Africa where malaria remains a leading cause of child mortality.
It also comes as part of a broader push that includes new vaccines, improved diagnostic tools and next-generation mosquito nets.
Novartis says it plans to supply the drug on a largely not-for-profit basis in high-burden countries, which could help speed up access in lower-income regions.
For many African families, the development could mean the difference between life and death in the earliest and most vulnerable months of a child’s life.